|
12/16/2023 0 Comments Solid or acuqeus precipitate chemistry  
The insoluble solid is called a precipitate and is. Referring to the solubility rules, MgCO 3 is insoluble in water resulting in a phase label of ( s), while NH 4Br is soluble in water resulting in a phase label of ( aq). In a precipitation reaction, two aqueous compounds react to form two compounds: one soluble, one insoluble. Students will analyze the chemical equation for the reaction and see that all atoms in the reactants end up in the products. Students will combine two clear colorless solutions (baking soda solution and calcium chloride solution) and see the formation of a solid and a gas. MgBr 2 ( aq) + (NH 4) 2CO 3 ( aq) → Mg CO 3 + NH 4 Br A solid formed from two solutions is called a precipitate. A may be by Chemistry experts to help you in doubts & scoring. Ammonium ions pair up with bromide ions forming NH 4Br (NH 4 + balances the charge of Br –). The aqueous solution gives a white precipitate. 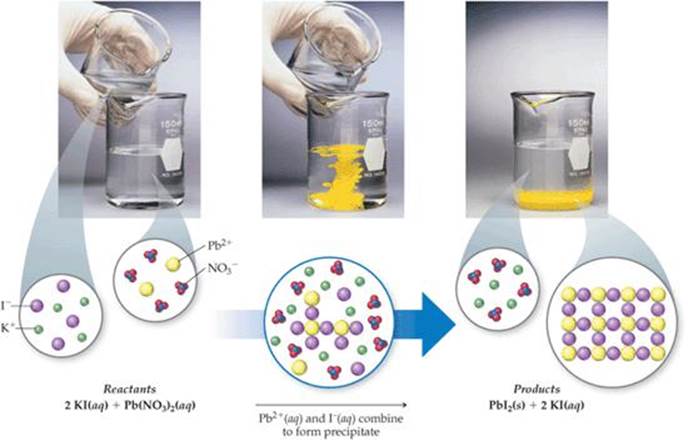
Magnesium ions pair up with carbonate ions forming magnesium carbonate, MgCO 3 (Mg 2 + balances the charge of CO 3 2 –). The chemical equation for this precipitation reaction between potassium chloride and silver nitrate is given below: \( AgNO3 (aqueous) + KCl (aqueous) \longrightarrow AgCl (precipitate) + KNO. Because silver chloride is a sparingly soluble salt, the equilibrium concentration of its dissolved ions in the solution is relatively low. MgBr 2 is called magnesium bromide and is comprised of magnesium ions, Mg 2 +, and bromide ions, Br −, while (NH 4) 2CO 3 is called ammonium carbonate and is comprised of ammonium ions, NH 4 +, and carbonate ions, CO 3 2 –. In this chemical reaction, the solid silver chloride is precipitated out, which is the insoluble salt formed as a product of the precipitation reaction. In this solution, an excess of solid AgCl dissolves and dissociates to produce aqueous Ag + and Cl ions at the same rate that these aqueous ions combine and precipitate to form solid AgCl (Figure 15.2). On the right hand side of the equation, the precipitant (AgCl) is written in its full formula and designated as a solid, since this is the precipitate that is. (OpenStax, CC BY 4.0, via Wikimedia Commons)Ĭomplete and balance the following equation. If no reaction takes place, write "No Reaction" after the arrow. \): An x-ray of the digestive organs of a patient who has swallowed a “barium milkshake.” A barium milkshake is a suspension of very fine BaSO 4 particles in water the high atomic mass of barium makes it opaque to x-rays. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
